Approval gives veterinarians a loop diuretic option to help manage pulmonary edema in dogs with congestive heart failure caused by myxomatous mitral valve disease (MMVD)
FORT WORTH, TEXAS (May 23, 2024) – Vetoquinol announces that UpCard®-CA1 (torsemide oral solution) received conditional approval from the U.S. Food & Drug Administration (FDA). UpCard-CA1 is a potent loop diuretic with a once daily administration to help manage pulmonary edema in dogs with congestive heart failure (CHF) caused by myxomatous mitral valve disease (MMVD).
“Diuretics are a cornerstone of managing acute and chronic CHF in dogs,” said Eric M. Alsup, DVM, General Manager, Vetoquinol USA. “A loop diuretic with once daily administration can help increase client compliance and patient comfort as veterinarians and owners manage a complex clinical syndrome like CHF.”1
UpCard-CA1 is available in an easily titrated oral solution for precise dosing adjustments. Torsemide (also known as torasemide) tablets have been approve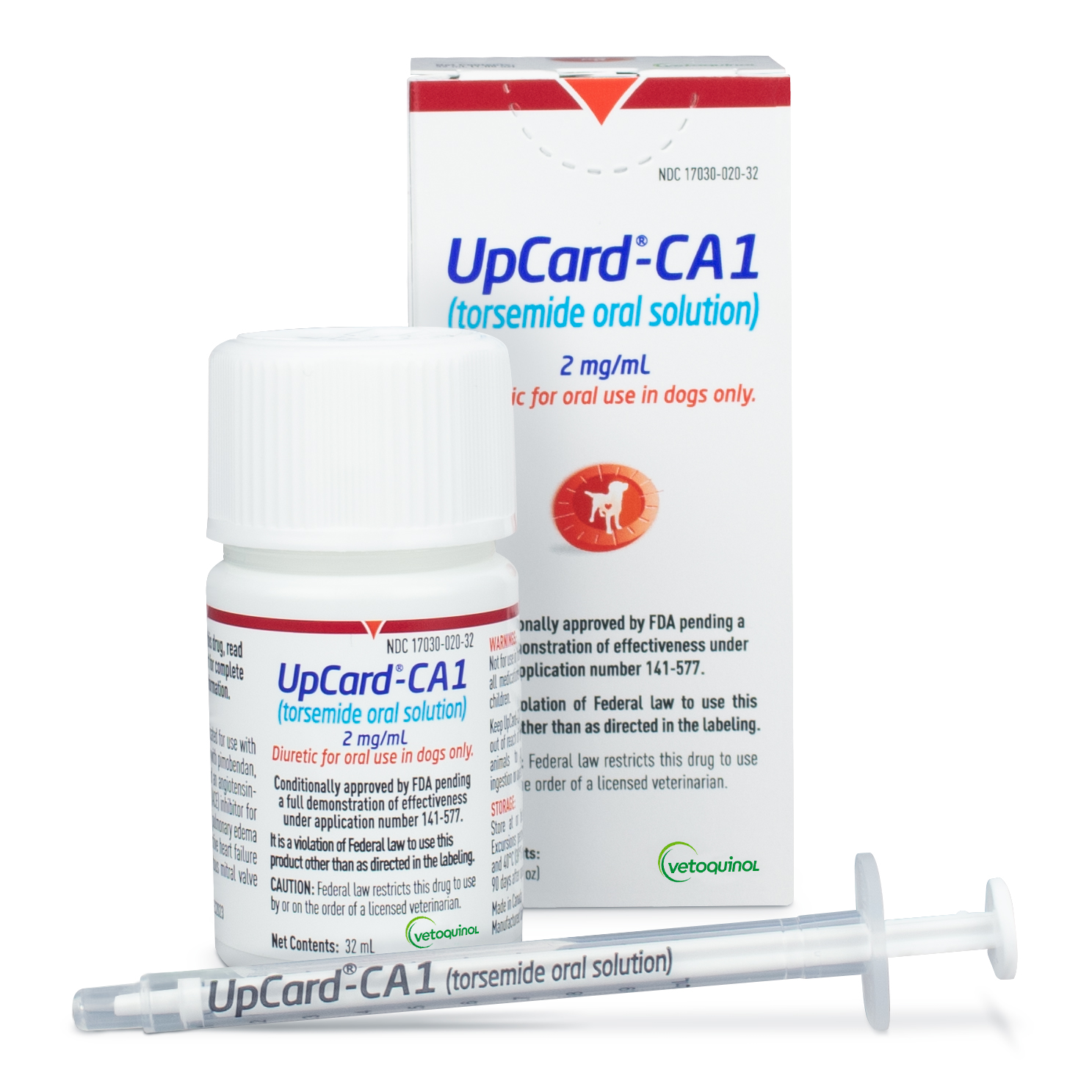 d for use as a veterinary diuretic in the European Union since 2015.
d for use as a veterinary diuretic in the European Union since 2015.
UpCard-CA1 is conditionally approved by the FDA pending a full demonstration of effectiveness under application number 141-577. A conditionally approved animal drug has gone through the FDA drug approval process except the drug has not yet met the effectiveness standard for full approval. FDA’s conditional approval means that, when used according to the label, the drug is safe and has a reasonable expectation of effectiveness.2
Veterinarians will be able to order UpCard-CA1 through distributors in July 2024.
UpCard-CA1 is conditionally approved by the FDA for use with concurrent therapy with pimobendan, spironolactone, and an angiotensin converting enzyme (ACE) inhibitor for the management of pulmonary edema in dogs with congestive heart failure caused by myxomatous mitral valve disease (MMVD).
About Vetoquinol USA
Headquartered in Fort Worth, Texas, Vetoquinol USA is owned by Vetoquinol S.A., an independent, family-owned French pharmaceutical company founded in 1933. Dedicated exclusively to animal health, Vetoquinol USA is focused on the development, production, and marketing of AAFCO, NASC, EPA, and FDA-regulated pharmaceutical, nutritional, and dermatological products for small and large animals.
IMPORTANT SAFETY INFORMATION:
UpCard-CA1 is for use in dogs only. Do not administer to dogs with renal failure, anuria, severe dehydration, hypovolemia, or hypotension. Do not administer UpCard-CA1 concomitantly with other loop diuretics or to dogs with hypersensitivity to the active substance, torsemide, or to any of the excipients. UpCard-CA1 should be used only in stable dogs with congestive heart failure caused by MMVD which has been diagnosed by means of a comprehensive physical and cardiac examination. This drug has not been evaluated in dogs used for breeding, pregnant or lactating bitches. The most common side effects seen in dogs with CHF due to MMVD while taking UpCard-CA1 are cough, dyspnea, pulmonary edema, and cardiac arrest. Adverse reactions not related to disease progression in dogs receiving UpCard-CA1 include polyuria and polydipsia, renal insufficiency, increased BUN and serum creatinine, urinary incontinence, hypokalemia, hypochloremia, hypercalcemia, hypomagnesemia, diarrhea, vomiting, and inappetence. For full prescribing information, visit www.vetoquinolusa.com/upcard-ca1-info.
For media requests:
usa_news@vetoquinol.com
For customer requests:
Contact Vetoquinol
817-529-7500
1. Chetboul, V., Pouchelon, J. L., Menard, J., Blanc, J., Desquilbet, L., Petit, A., Rougier, S., Lucats, L., Woehrle, F., & TEST study investigators (2017). Short-Term Efficacy and Safety of Torasemide and Furosemide in 366 Dogs with Degenerative Mitral Valve Disease: The TEST Study. Journal Of Veterinary Internal Medicine, 31(6), 1629–1642.
2. U.S. Food & Drug Administration. (2019). Unapproved Animal Drugs. https://www.fda.gov/animal-veterinary/compliance-enforcement/unapproved…. [Access date: April 12, 2024]
©2024 VETOQUINOL USA, Inc. VETOQUINOL and UPCARD are trademarks of VETOQUINOL.
Latest news

November 2025

Vetoquinol, Expert Educators Join Forces to Create Online Veterinary Rehabilitation Courses
April 2025
FDA Approves Extended Expiration Date for Clevor® (ropinirole ophthalmic solution)
August 2024
